
Low Coordinate Germanium(II) and Tin(II) Hydride Complexes: Efficient Catalysts for the Hydroboration of Carbonyl Compounds | Chem-Station Int. Ed.

Molecules | Free Full-Text | Synthesis, Characterization, and Reaction Studies of Pd(II) Tripeptide Complexes | HTML

Bulky N‐Heterocyclic‐Carbene‐Coordinated Palladium Catalysts for 1,2‐Addition of Arylboron Compounds to Carbonyl Compounds - Okuda - 2020 - ChemCatChem - Wiley Online Library

Umpolung of Carbonyl Groups as Alkyl Organometallic Reagent Surrogates for Palladium‐Catalyzed Allylic Alkylation - Zhu - 2018 - Angewandte Chemie - Wiley Online Library

CO Surrogates: A Green Alternative in Palladium-Catalyzed CO Gas Free Carbonylation Reactions | Bentham Science

Palladium nanoparticles on amino-modified silica-catalyzed C–C bond formation with carbonyl insertion | SpringerLink

One‐Pot Syntheses of α,β‐Unsaturated Carbonyl Compounds through Palladium‐Mediated Dehydrogenation of Ketones, Aldehydes, Esters, Lactones and Amides - Muzart - 2010 - European Journal of Organic Chemistry - Wiley Online Library

Palladium‐Catalyzed Selective Reduction of Carbonyl Compounds - Sarkar - 2020 - European Journal of Inorganic Chemistry - Wiley Online Library
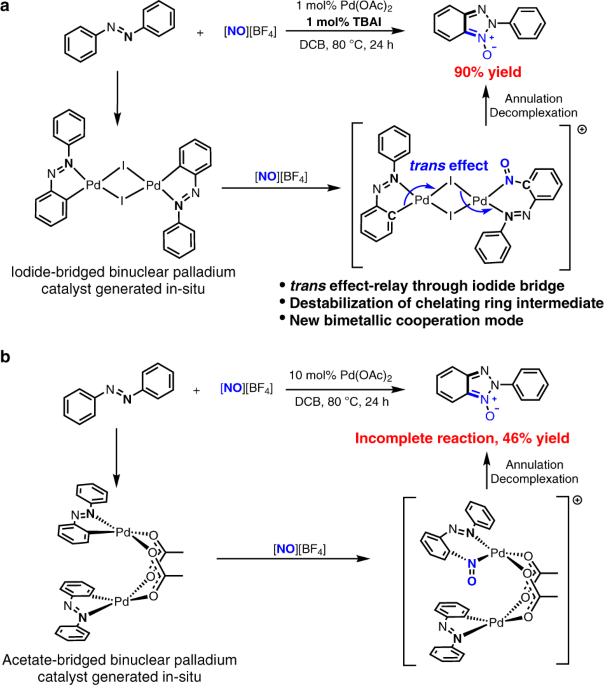
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex | Communications Chemistry
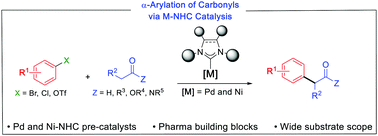
N-Heterocyclic carbene complexes enabling the α-arylation of carbonyl compounds - Chemical Communications (RSC Publishing)
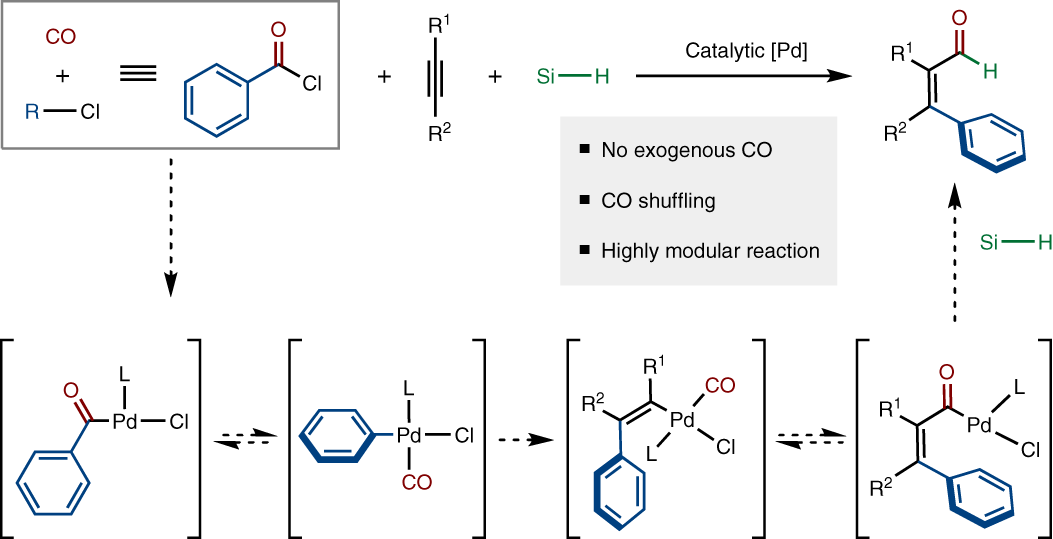
Palladium-catalysed carboformylation of alkynes using acid chlorides as a dual carbon monoxide and carbon source | Nature Chemistry

18. M(0) carbonyl complexes with chelating di-NHC or tri-NHC ligands... | Download Scientific Diagram

Industrial catalyst | Palladium(+1) Carbonyl Clusters in the Catalytic Oxidation of Unsaturated Compounds

Pyrazol-1-yl)carbonyl palladium complexes as catalysts for ethylene polymerization reaction - ScienceDirect

Scheme 20. Carbonylation of hydroxide pincer complexes 12a and 12b. The... | Download Scientific Diagram
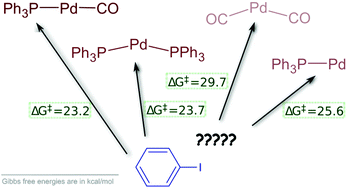
Viable pathways for the oxidative addition of iodobenzene to palladium (0)-triphenylphosphine-carbonyl complexes: a theoretical study - Dalton Transactions (RSC Publishing)

Diastereoselective Carbonyl Allylation with Simple Olefins Enabled by Palladium Complex-Catalyzed C-H Oxidative Borylation

Binuclear Palladium Complex Immobilized on Mesoporous SBA-16: Efficient Heterogeneous Catalyst for the Carbonylative Suzuki Coupling Reaction of Aryl Iodides and Arylboronic Acids Using Cr(CO)6 as Carbonyl Source | SpringerLink

![16. M(0) di-NHC carbonyl complexes [72,81,99,100,109-111]. | Download Scientific Diagram 16. M(0) di-NHC carbonyl complexes [72,81,99,100,109-111]. | Download Scientific Diagram](https://www.researchgate.net/profile/Lu-Jiang-5/publication/272102102/figure/fig5/AS:572844640620545@1513588213405/M0-di-NHC-carbonyl-complexes-72-81-99-100-109-111.png)



![20. M(0) carbonyl complexes with hybrid NHC ligands [114,120-122]. | Download Scientific Diagram 20. M(0) carbonyl complexes with hybrid NHC ligands [114,120-122]. | Download Scientific Diagram](https://www.researchgate.net/profile/Lu-Jiang-5/publication/272102102/figure/fig8/AS:572844641271822@1513588213512/M0-carbonyl-complexes-with-hybrid-NHC-ligands-114-120-122_Q640.jpg)
